Security Testing That Gets Medical Devices to Market
Penetration testing and threat modeling for medical device manufacturers — from FDA submission through post-market. We test your device in our lab and deliver actionable, FDA-ready reports.
Our Services
Penetration Testing
Hands-on security testing of your device in our hardware lab — network protocols, firmware, physical interfaces, and cloud components.
$45,000Threat Modeling
Structured analysis of your device's architecture, attack surfaces, and risk profile. Identifies vulnerabilities before testing begins.
$15,000FDA ANIN Response
Received an ANIN cybersecurity letter from the FDA? We help you address deficiencies, conduct the required testing, and resubmit with confidence.
Equivalency Validation
Cybersecurity testing for substantial equivalence claims. We validate that your device meets the same security posture as the predicate device in your submission.
Custom Engagements
Unique device or testing scope? We'll define a custom engagement with tailored pricing.
Contact Us →FDA-Compliant Reports — Included with Every Engagement
Every engagement produces a report mapped to FDA premarket cybersecurity guidance, ready to include in your 510(k) or PMA submission. Executive summary, methodology, findings with proof-of-concept, risk assessment, and remediation roadmap.
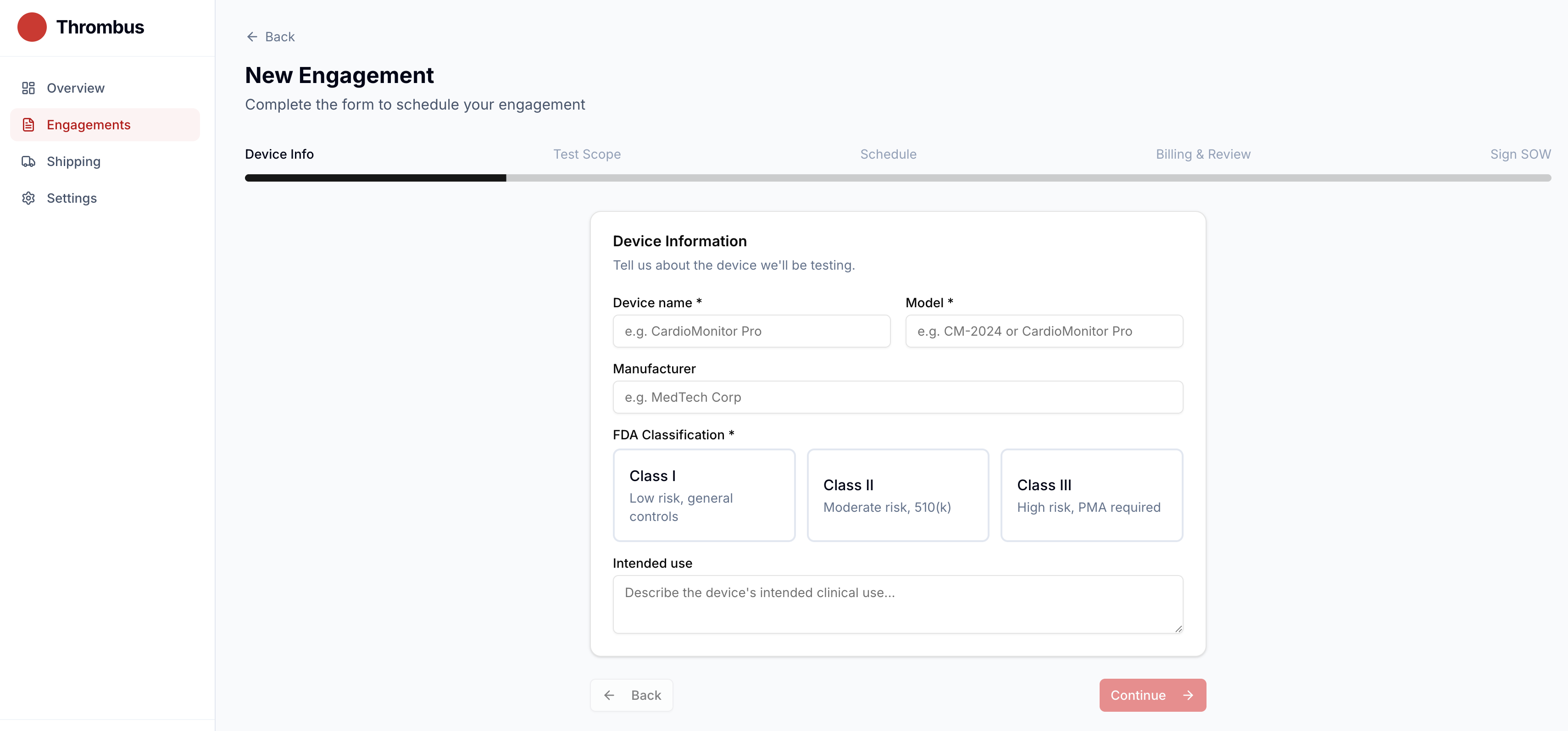
Manage Your Pentest Engagement Online
Thrombus is our pentest management platform. Create an engagement, ship your device, track progress, review findings, download your FDA-compliant report, and pay — all in one place.
- Transparent, upfront pricing
- Real-time engagement status tracking
- Secure document sharing
- FDA-compliant report delivery
Latest Articles

It Was Just Cached Credentials — And It Triggered an FDA Recall
GE HealthCare's Centricity Universal Viewer was recalled in March 2026 over exposed login credentials on a local workstation. No remote exploit, no zero-day — and that's exactly why every device manufacturer should pay attention.
Hardware Security in Medical Devices: What We Find in the Lab
A look at the most common hardware security issues we encounter when testing medical devices — from exposed debug ports to unencrypted flash storage. What manufacturers miss and how to fix it.
Your security partner, not just a vendor
Every engagement lives in our platform, giving you full visibility from kickoff to final report and beyond. We're here for every device, every year.